Tick maps
The tick maps are published regularly to provide the ECDC stakeholders, the scientific community and the general public with updated information on tick distribution at ‘regional’ administrative unit level (NUTS3 or equivalent and NUTS2 in Austria, Belgium, Denmark, Germany, The Netherlands, England (UK), Scotland (UK) and Wales (UK)).
Please note that the maps do not represent the official view or position of the countries.
Distribution maps of ticks
The map shows the current known distribution of the tick species in Europe at ‘regional’ administrative level (NUTS3). They are based on published historical data and confirmed data provided by experts from the respective countries as part of the VectorNet project.
Separate maps for the distribution of each of the following tick species are currently available: Dermacentor reticulatus, Hyalomma lusitanicum, Hyalomma marginatum, Ixodes persulcatus, Ixodes ricinus, Ornithodoros erraticus, Rhipicephalus sanguineus.
For native species the possible distribution statuses are:
- Present: The species has been observed to be present in at least one municipality within the administrative unit.
- Introduced: The species has been introduced in the administrative unit without confirmed establishment.
- Anticipated Absent: the species has never been reported and there is a high probability that it is absent.
- Confirmed Absent: the species has never been reported within the administrative unit and there have been field surveys or studies on ticks within the last 5 years of the distribution status date.
- No Data: No sampling has been performed and no data on the species is available.
- Unknown: The status is unknown
Areas outside the scope of the VectorNet geographical scope are colour-coded as ‘Outside scope’.
Vector distribution status changes in terms of spatial units since the previous update
Since the previous update (August 2023), updates for the seven species are as follows:
- Dermacentor reticulatus: data captured for 5 NUTS3s, with no changes suggested for any NUTS3s
- Hyalomma lusitanicum: data captured for 6 NUTS3s, with no changes suggested for any NUTS3s
- Hyalomma marginatum: data captured for 8 NUTS3s, and changes suggested for 2 NUTS3 – including new areas of presence in Algeria and Tunisia
- Ixodes persulcatus: no new data captured for this reporting period
- Ixodes ricinus: data captured for 128 NUTS3s, and changes suggested for 3 NUTS3s – including new areas of presence in Croatia, France and Spain
- Ornithodoros erraticus: no new data captured for this reporting period
- Rhipicephalus sanguineus: data captured for 10 NUTS3s, and changes suggested for 4 NUTS3s – including new areas of presence in Spain (northern Spain and Tenerife) and Western Sahara.
Dermacentor reticulatus
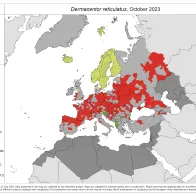
61 new reports were submitted since the previous update (August 2023).
Hyalomma lusitanicum
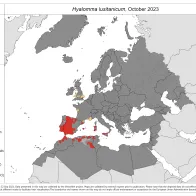
6 new reports were submitted since the previous update (August 2023).
Hyalomma marginatum
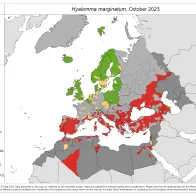
32 new reports were submitted since the previous update (March 2023).
Ixodes persulcatus
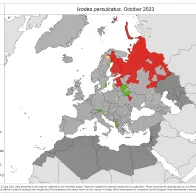
0 new reports were submitted since the previous update (March 2023).
Ixodes ricinus
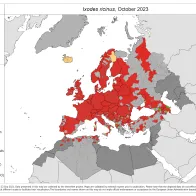
1057 new reports were submitted since the previous update (August 2023).
Ornithodorus erraticus
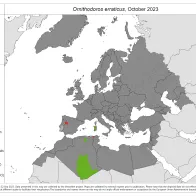
0 new reports were submitted since the previous update (August 2023).
Rhipicephalus sanguineus
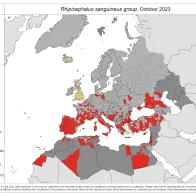
61 new reports were submitted since the previous update (August 2023).
Surveillance for ticks
The surveillance maps shows the regions where surveillance activities for ticks were held in Europe and neighbouring regions.
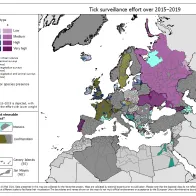
The map Tick surveillance effort over 2015–2019' shows the surveillance effort per administrative area, by highest surveillance type.
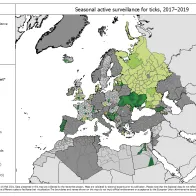
The map 'Seasonal active surveillance for ticks over 2017–2019' shows the number of different calendar months in which any active surveillance took place per administrative area.