European Antimicrobial Resistance Genes Surveillance Network (EURGen-Net)
The European Antimicrobial Resistance Genes Surveillance Network (EURGen-Net) is a network for genomic-based surveillance of multidrug-resistant bacteria of public health importance, coordinated by the European Centre for Disease Prevention and Control (ECDC). National reference laboratories or equivalent laboratories of 37 European countries currently participate in EURGen-Net.
The public health objectives of this European whole genome sequencing (WGS)-based surveillance are to determine the geographic distribution and population dynamics of multidrug-resistant clones and transmissible resistance elements to inform risk assessment, prevention and control polices and to support countries in developing technical capability and proficiency for genomic-based surveillance of multidrug-resistant bacteria with epidemic potential.
Survey of carbapenem-resistant Acinetobacter baumannii in Europe
ECDC launched in July 2024 a protocol for a genomic survey of carbapenem-resistant Acinetobacter baumannii (CRAb) across hospitals from EU/EEA countries, Western Balkan countries and Türkiye.
The protocol describes actions for hospitals and clinical microbiology laboratories to conduct a survey that aims to map the occurrence, geographic distribution and resistance characteristics of CRAb isolates.
The survey also aims to help countries enhance their capacities for detecting and controlling infections caused by CRAb. This includes strengthening national capabilities for implementing advanced genetic techniques to monitor CRAb and understanding the factors that lead to CRAb infections.
The survey protocol will be accompanied by a laboratory manual aimed at professionals working in clinical laboratories, including reference laboratories.
Once completed, the survey results will inform national and European CRAb preparedness, prevention and control activities.
The survey, conducted through ECDC’s European Antimicrobial Resistance Genes Surveillance Network (EURGen-Net), will take place between October 2024 and June 2025.
Protocols for CRAb survey
The survey of carbapenem-resistant Enterobacterales 2025 (CRE25 survey)
The survey of carbapenem-resistant Enterobacterales 2025 (CRE25 survey) is the third European survey of carbapenem-resistant Enterobacterales following the European survey of carbapenemase-producing Enterobacteriaceae (EuSCAPE) in 2013-2014 and the survey of carbapenem- and/or colistin-resistant Enterobacterales (CCRE survey) in 2019. The CRE25 survey follows a similar protocol to the two previous surveys but with several modifications to allow for faster completion of data collection and analysis of results.
For the CRE25 survey, NRLs from the 37 countries participating in EURGen-Net are invited to submit WGS data to ECDC accompanied by standardised epidemiological and microbiological metadata for up to 40 isolates per Nomenclature of Territorial Units for Statistics level 2 (NUTS-2) region, according to the inclusion criteria outlined in the protocol. For NRLs without sufficient WGS capacity, central sequencing support will be provided.
Protocols for CRE25
Europe-wide survey of carbapenem- and/or colistin-resistant Enterobacteriaceae (CCRE survey)
The CCRE survey complements the phenotypic antimicrobial resistance data available from the European Antimicrobial Resistance Surveillance Network (EARS-Net) with genomic information to identify high-risk clones and gain a more detailed understanding of transmission pathways. The CCRE survey results will be used at national and European level for risk assessment and decision-making as well as better targeting control measures.
During a six-month period, hospitals and their associated clinical microbiology laboratories in 37 European countries are collecting carbapenem- and/or colistin-resistant Klebsiella pneumoniae and Escherichia coli isolates, as well as corresponding susceptible comparator isolates, based on a scheme that takes into account the countries’ population and geographic structure. The CCRE survey includes a multi-step process starting with collection of bacterial isolates at local hospital level, followed by forwarding of the isolates to the national reference laboratory for confirmatory testing and finally sending the isolates to a central strain collection for subsequent WGS and related analysis. Up to 6000 isolates are expected to be collected and analysed for this CCRE survey.
Results

Protocols for the CCRE survey
EURGen-Net publications
Investigation of the spread of carbapenem-resistant Klebsiella pneumoniae in Greek hospitals
Investigation of New Delhi metallo-beta-lactamase (NDM)-1-producing Providencia stuartii in hospitals in Romania
Investigation of NDM-5 producing Escherichia coli in the EU/EEA
Related publications
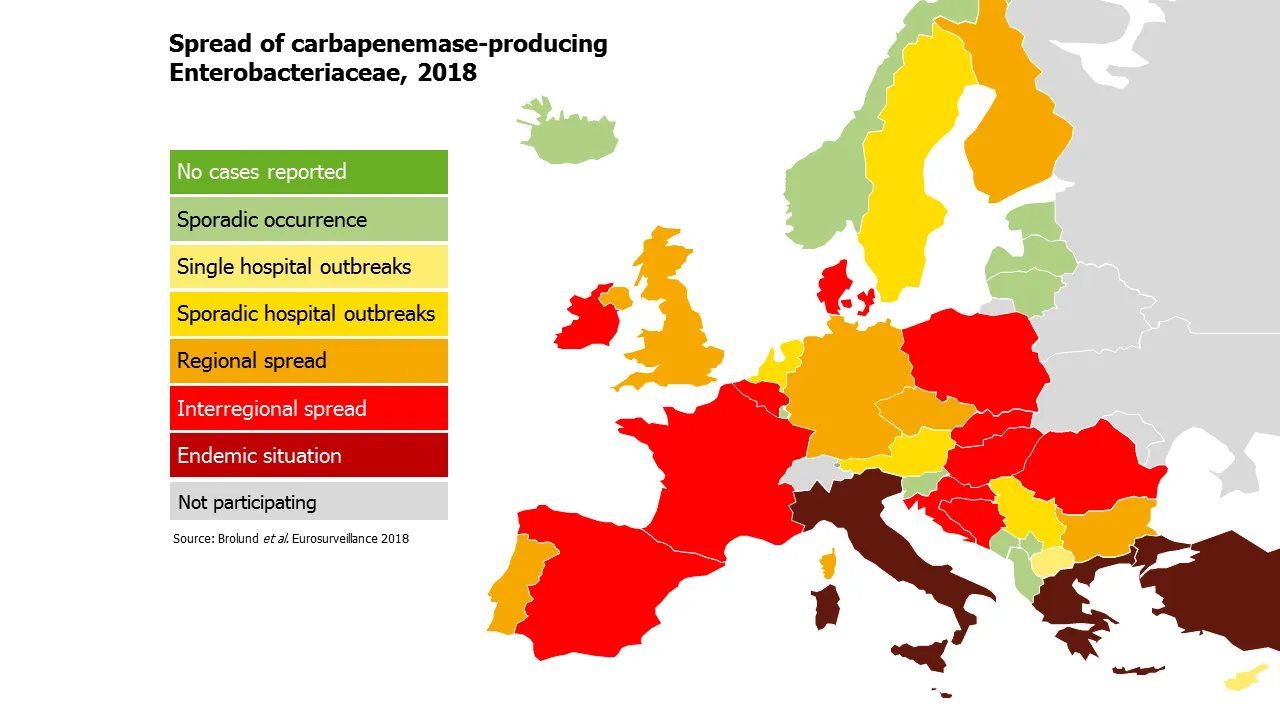
European survey of carbapenemase-producing Enterobacteriaceae (EuSCAPE), 2013-2015
In 2012, the European Centre for Disease Prevention and Control (ECDC) launched the “European survey of carbapenemase-producing Enterobacteriaceae” (EuSCAPE) project. This project demonstrated the feasibility of structured surveys of carbapenemase-producing Enterobacteriaceae at the European level, raised awareness about their spread, and contributed to improving the corresponding laboratory and response capacity in European countries.

Related ECDC publications



Monitoring the use of whole-genome sequencing in infectious disease surveillance in Europe 2015–2017