Disease vectors
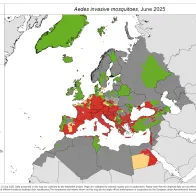
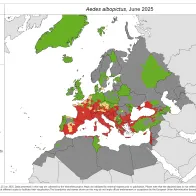
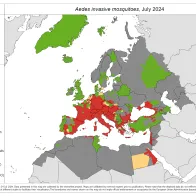
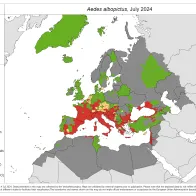
Vector borne diseases are a specific group of infectious diseases that are a (re-)emerging threat to Europe. One important aspect of preparedness for vector borne diseases is the surveillance of the introduction, establishment and spread of the main disease vectors.
Through VectorNet, ECDC maintains a database on the presence and distribution of vectors in Europe and regularly publishes updated distribution maps.