Pre-exposure prophylaxis in the EU/EEA. PrEP service delivery and monitoring: minimum standards and key principles
HIV pre-exposure prophylaxis is the use of antiretroviral medicines to prevent HIV among people who are HIV-negative. It effectively prevents HIV infection when taken as prescribed, e.g. during times when the likelihood of becoming infected with HIV is very high, or, for gay men and other men who have sex with men, before and after sexual activity, so-called event-driven PrEP.
Observations and findings
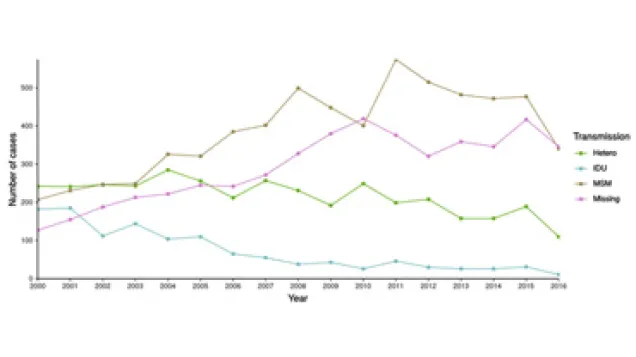
Several cities across Europe where PrEP is offered have seen a drop in the number of HIV diagnoses among gay men and other men who have sex with men. But overall, PrEP access and uptake in Europe is not yet sufficient to affect the overall course of the HIV epidemic across Europe.
Sex between men is the main HIV transmission mode in the EU/EEA and PrEP users in these countries are almost all gay men and other men who have sex with men. But the vast majority of those at risk of HIV do not have formal access to PrEP. Consequently, among those taking PrEP, many purchase it online and take it with no medical support and monitoring.
During the meeting, several modes of PrEP provision where presented. The offer of PrEP in friendly and accessible settings can attract people to HIV testing as well as for diagnosis and treatment of other sexually transmitted infections. Taking PrEP provision out of highly specialised centres, in consultation with communities of potential users, also provides a gateway to broader healthcare. Innovative online approaches to improve PrEP self-management and sexually transmitted infection diagnosis and treatment were also presented.
Maturing European PrEP programmes are leading on ways to achieve the equitable and rapid scale-up of this intervention that is needed to make an impact on the HIV epidemic in Europe. Decentralisation of PrEP provision and integration with broader health services often has to be achieved without an increase in funding. Service providers are therefore looking for guidance on minimum safe standards for PrEP delivery.
Users and providers alike among the meeting participants agreed that the patterns of use are still evolving, with no clear distinction between daily and event-driven PrEP. Factors such as the ability to afford the next PrEP prescription, and improved identification of times when there will be a high chance of acquiring HIV, are increasingly determining how this medication is being taken.
Taking it incorrectly, can result in HIV infection or lead to the development of resistance to the antivirals. Therefore, the meeting participants called for guidelines on how to effectively start and stop PrEP, HIV and STI testing regimens to follow for this type of non-daily use to be safe.
Three actions were identified during the meeting:
-
A consultation paper will be developed on essential and desirable components and principles of a decentralised PrEP programme, including safe non-daily use.
-
A standardised monitoring tool will be developed by ECDC in 2019.
-
Meeting attendees will be asked to give what details they can on pricing of PrEP medicines to governments and consumers, which will be shared between European HIV programmes in order to strengthen price negotiations.


All updates on HIV/AIDS
Featured
Share this page