Invasive meningococcal disease - Annual Epidemiological Report 2016 [2014 data]
invasive meningococcal disease - Annual Epidemiological Report for 2014
English (177.61 KB - PDF)Key facts
- In 2014, 2 760 confirmed cases of invasive meningococcal disease were reported to TESSy.
- The notification rate was 0.5 cases per 100 000 population, which is lower than in previous years.
- Age-specific rates were highest in infants (10.1 cases per 100 000 population), followed by 1–4-year-olds (2.5 cases per 100 000 population).
- The majority of cases were caused by serogroup B (64%).
- Serogroup C is more prominent in countries that do not have the meningococcal C conjugate (MCC) vaccine in their routine national immunisation schedules.
- Continued strengthening of disease surveillance for invasive meningococcal is essential to evaluate the impact of ongoing immunisation programmes and to support decision-makers in view of the availability of new vaccines.
Methods
Click here for a detailed description of the methods used to produce this annual report
ECDC has coordinated the surveillance of invasive meningococcal disease (IMD) at the European level since the transfer of EU-IBIS (European Union Invasive Bacterial Infections Surveillance Network) to ECDC in 2007.
- In 2014, data were reported by 30 Member States.
- In 2014, 11 Member States used the EU-2012 case definition (Commission Implementing Decision 2012/506/EU of 8 August 2012), 14 Member States used the EU-2008 definition, and one Member State used the EU-2002 case definition. Four Member States used alternative case definitions.
- Surveillance systems used by Member States for reporting data on IMD to ECDC are heterogeneous, with differences in the type of system in place, population coverage of the system, and method of data reporting. For example, some systems are based on clinical syndromes such as meningitis while other systems rely exclusively on laboratory results, with the number of performed blood cultures varying. Surveillance systems in Member States and at the European level also change and develop over time, for example in respect to representativeness and reporting procedures.
- All national surveillance systems provide full national coverage, and all countries – except for Bulgaria and Croatia – report case-based data (see Annex).
Epidemiology
In 2014, 2 760 confirmed cases of IMD were reported by 30 EU/EEA countries. The notification rate was 0.5 cases per 100 000 population (0.7 cases per 100 000 population in both 2012 and 2013; Table 1, Figure 1). The highest notification rates were observed in Malta (3.1 cases per 100 000 population), Lithuania (1.8), Ireland (1.7) and the UK (1.2) (Table 1, Figure 2).
Age and gender distribution
In 2014, IMD was predominantly found in infants and young children (Figure 3), with a notification rate of 10.1 confirmed cases per 100 000 population in children under one year of age, and 2.5 confirmed cases per 100 000 population in 1–4-year-olds. When stratifying by gender, there was a slight predominance of cases in males versus females in the age groups under 45 years (Figure 3). The overall male-to-female ratio was 1.1:1.
Seasonality
In 2014, seasonality followed a pattern similar to previous years. IMD occurred primarily in the winter months, while the number of cases was lowest in summer (Figures 4 and 5). The monthly number of cases in 2014 was consistently lower than the mean number of cases in 2010–2013 (Figure 4). Overall, a declining trend in the number of IMD cases could be observed (Figure 5).
Serogroup
Of the 2 760 IMD cases reported in 2014, the serogroup was known for 2 456 (89%). Of the cases with known serogroup, the majority belonged to serogroup B (64%), followed by serogroup C (16%) (Table 2). There has been a decrease in serogroup B since 2011 and in serogroup C since 2012. Serogroup W has been increasing since 2011, while serogroup Y has stayed relatively stable (Figure 6). Serogroup B accounted for more than 80% of IMD in children under the age of five years, but only for 31% of cases aged 65 years and over. Serogroup C was most prominent in 25–44-year-olds, accounting for 30% of cases in this age group. Serogroups Y and W were most prominent in those aged 65 years and over, causing 30% and 18% of IMD cases in this age group, respectively (Figure 7).
By 2014, 14 EU countries had integrated the meningococcal C conjugate (MCC) vaccine into their routine national childhood immunisation programmes. In countries that have not implemented MCC vaccination, serogroup C accounted for 27% of cases, while in countries that have implemented MCC vaccination, only 14% of the cases were attributed to serogroup C (Figure 7).
Clinical presentation
The clinical presentation was known for 1 351 cases (49%). Meningitis was reported in 552 cases (41%), septicaemia in 462 cases (34%). Both septicaemia and meningitis were reported in 232 cases (17%). The remaining 105 cases (8%) showed a different clinical presentation.
Outcome
The outcome was known for 2 370 cases, 86% of all cases. Among cases with known outcome, 214 were fatal (case fatality: 9%). Case fatality was highest among cases of serogroup C (15%, n=55/355) and lowest among cases of serogroup B (7%, n=95/1 364). Case fatality was highest in cases aged 65 years and over (14%, n=42/300), and lowest in 5–14-year-olds (6%, n=14/234).
Discussion
Invasive meningococcal disease remains rare in EU/EEA countries, with the greatest burden in infants and young children. In 2014, country-specific notification rates ranged from 0.2 to 3.1 cases per 100 000 population. Although IMD is rare, it is a severe and life-threatening disease with relatively high case fatality and with up to one fifth of all survivors suffering from long-term sequelae [1].
There continues to be a decreasing trend in serogroup B, which may be due to a naturally occurring secular trend [2]. However, serogroup B continues to cause the majority of cases of IMD, predominantly affecting the younger age groups. In Europe, a recombinant protein vaccine including outer membrane vesicles against serogroup B (4CMenB) was licensed in 2013 and was estimated to provide protection against between 73% and 87% of circulating serogroup B strains, depending on the country [3], as well as potential cross protection against IMD caused by other serogroups [4, 5]. The United Kingdom introduced 4CMenB into its routine national childhood immunisation programme in 2015 [6]. Ireland is scheduled to introduce the vaccine into its routine immunisation programme in September 2016. The vaccine is recommended, but not funded, in Austria, the Czech Republic and Hungary. Following assessment, some countries, such as France, Greece and Norway, recommended that the 4CMenB vaccine should not be introduced into their routine immunisation programmes. However, in France the vaccine is recommended for persons at increased risk of IMD and during outbreaks.
Since 1999, 14 EU/EEA countries have introduced MCC vaccination into their routine national childhood immunisation programme [6], and the impact of MCC vaccination has been well demonstrated [2, 7-10]. In Europe, serogroup C continues to show a decreasing trend and is proportionally twice as prominent in countries that do not include MCC vaccination in their routine national childhood immunisation programme.
Several EU/EEA countries have reported increasing trends in serogroup Y in recent years [2, 9, 11], although this was not observed at the EU level during the time period presented in this report. Serogroup W has been increasing at the European level since 2011, although this increase is solely related to the rapid epidemic expansion of a single clone in the UK that started in 2009–2010 [12]. In recent years, four countries (the UK, Greece, Austria and the Czech Republic) have introduced the quadrivalent conjugate vaccine MenACYW into their routine vaccination schedules, predominantly as booster doses for adolescents [6]. As carriage rates of Neisseria meningitidis are highest in adolescents and young adults [13], high levels of immunity in this age group are critical to ensure the protection of other vulnerable age groups [14].
Public health conclusions
Several vaccines targeting different serogroups exist for the prevention of invasive meningococcal disease. The choice of introducing a vaccine into the routine national immunisation programme depends on the disease and vaccine attributes, as well as context-specific factors in each country, such as the disease and serogroup burden, cost-effectiveness and feasibility.
Continued strengthening of IMD surveillance is essential to evaluate the impact of ongoing immunisation programmes and to support decision-makers in view of the availability of new vaccines. Surveillance at the European level will become even more important as the incidence of the disease declines, and the pooling of data may enable the description of trends which are difficult to discern at the national level.
References
- Rosenstein NE, Perkins BA, Stephens DS, Popovic T, Hughes JM. Meningococcal disease. N Engl J Med. 2001;344(18):1378-88
- Bijlsma MW, Bekker V, Brouwer MC, Spanjaard L, van de Beek D, van der Ende A. Epidemiology of invasive meningococcal disease in the Netherlands, 1960-2012: an analysis of national surveillance data. Lancet Infect Dis. 2014;14(9):805-12.
- Vogel U, Taha MK, Vazquez JA, Findlow J, Claus H, Stefanelli P, et al. Predicted strain coverage of a meningococcal multicomponent vaccine (4CMenB) in Europe: a qualitative and quantitative assessment. Lancet Infect Dis. 2013;13(5):416-25.
- Trotter CL, Ramsay ME. Vaccination against meningococcal disease in Europe: review and recommendations for the use of conjugate vaccines. FEMS Microbiol Rev. 2007 Jan;31(1):101-7.
- Wise J. Teenagers in England to be vaccinated against meningitis group W. BMJ. 2015 Mar 16;350:h1486.
- European Centre for Disease Prevention and Control. Vaccine scheduler. [Internet.] Stockholm: ECDC; 2016. Available at: Vaccine Scheduler | ECDC (europa.eu).
- Borrow R, Abad R, Trotter C, van der Klis FR, Vazquez JA. Effectiveness of meningococcal serogroup C vaccine programmes. Vaccine. 2013 Sep 23;31(41):4477-86.
- Garrido-Estepa M, León-Gómez I, Herruzo R, Cano R. Changes in meningococcal C epidemiology and vaccine effectiveness after vaccine introduction and schedule modification. Vaccine. 2014 May 7;32(22):2604-9.
- Hellenbrand W, Elias J, Wichmann O, Dehnert M, Frosch M, Vogel U. Epidemiology of invasive meningococcal disease in Germany, 2002–2010, and impact of vaccination with meningococcal C conjugate vaccine. J Infect. 2013 Jan;66(1):48-56.
- Larrauri A, Cano R, García M, Mateo Sd. Impact and effectiveness of meningococcal C conjugate vaccine following its introduction in Spain. Vaccine. 2005 Jul 14;23(32):4097-100.
- Bröker M, Emonet S, Fazio C, Jacobsson S, Koliou M, Kuusi M, et al. Meningococcal serogroup Y disease in Europe continuation of high importance in some European regions in 2013. Hum Vaccin Immunother. 2015;11(9):2281-6.
- Ladhani SN, Beebeejaun K, Lucidarme J, Campbell H, Gray S, Kaczmarski E, et al. Increase in endemic Neisseria meningitidis capsular group W sequence type 11 complex associated with severe invasive disease in England and Wales. Clin Infect Dis. 2015 Feb 15;60(4):578-85.
- Cohn AC, MacNeil JR, Clark TA, Ortega-Sanchez IR, Briere EZ, Meissner HC, et al. Prevention and control of meningococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2013 Mar 22;62(RR-2):1-28.
- Maiden MC, Ibarz-Pavón AB, Urwin R, Gray SJ, Andrews NJ, Clarke SC, et al. Impact of meningococcal serogroup C conjugate vaccines on carriage and herd immunity. J Infect Dis. 2008 Mar 1;197(5):737-43.
Additional information
- ECDC Surveillance Atlas of Infectious Diseases
- Network background and EU-IBIS reports: http://www.ecdc.europa.eu/en/activities/surveillance/EU_IBD/background/Pages/Background.aspx
_________
* The European Surveillance System (TESSy) is a system for the collection, analysis and dissemination of data on communicable diseases. EU Member States and EEA countries contribute to the system by uploading their infectious disease surveillance data at regular intervals.
Reported confirmed cases of invasive meningococcal disease cases: number and rate per 100 000 population, EU/EEA, 2010–2014
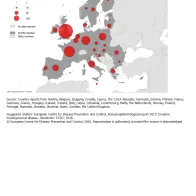
Figure 1. Reported confirmed cases of invasive meningococcal disease, EU/EEA, 2014

Number of cases of Reported confirmed invasive meningococcal disease per 100 000 population, EU/EEA, 2014
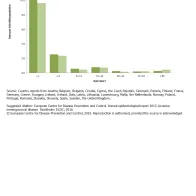
Reported confirmed cases of invasive meningococcal disease cases, by age and gender, EU/EEA, 2014
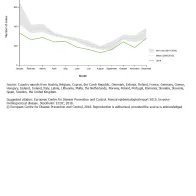
Seasonal distribution of Reported confirmed cases of invasive meningococcal disease, EU/EEA, 2014 compared with 2010−2013

Trend and number of cases of Reported confirmed invasive meningococcal disease, EU/EEA, 2010−2014
Serogroup distribution of confirmed cases of invasive meningococcal disease, EU/EEA, 2014

Figure 6. Trend of confirmed cases of invasive meningococcal disease by serogroup, EU/EEA, 2010–2014
Trend of confirmed cases of invasive meningococcal disease by serogroup, EU/EEA, 2010–2014

Serogroup distribution of confirmed cases of invasive meningococcal disease by age group, EU/EEA, 2014
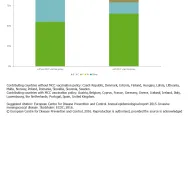
Trend and number of cases of reported, confirmed invasive meningococcal disease, EU/EEA, 2010-2014
Table containing overview of surveillance systems for Invasive meningococcal disease in 2014
Share this page
