EU Laboratory Capability Monitoring System (EULabCap): Report on 2015 survey of EU/EEA country capabilities and capacities
Executive Summary
The ECDC public health microbiology strategy (2012–2016) aims to strengthen the capability and capacity of the EU public health microbiology system to provide the timely and reliable information that underpins infectious threat detection, assessment and surveillance at EU and Member State levels to prevent and control infectious diseases. To ascertain how well this is delivered, ECDC has developed the EULabCap system for monitoring key public health microbiology capabilities and capacity for EU surveillance and epidemic preparedness.
EU Laboratory Capability Monitoring System (EULabCap): Report on 2015 survey of EU/EEA country capabilities and capacities
English (2.33 MB - PDF)Related maps
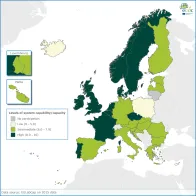
The average EULabCap aggregated index score was 7.5 on a scale of 0-10 in 2016 for the 30 participating EU/EEA countries
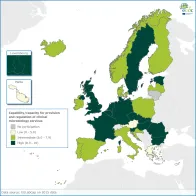
Provision of reliable, quality-assured, safe and fully-accessible clinical diagnostic microbiology services is a prerequisite for adequate case ascertainment and surveillance/threat notification systems.
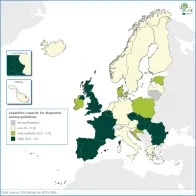
Availability of national primary diagnostic and screening testing guidelines (e.g. who to test, how to test, and when to test) is a prerequisite to guarantee sufficient sensitivity for case ascertainment and surveillance/threat notification systems.

Awareness of national testing practices provides a basis for monitoring sensitivity of case ascertainment and surveillance/notification systems.
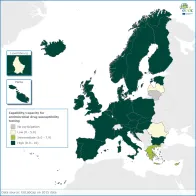
Implementation and monitoring of compliance with EU standards for antimicrobial drug susceptibility testing is a prerequisite for accurate and comparable EU surveillance of antimicrobial resistance, in accordance with EU strategy on AMR.

Organisation, regulation, and funding of national reference laboratory infrastructure and core public health functions are key elements for informing surveillance and epidemic preparedness at national and EU levels, in accordance with NMFP consensus.
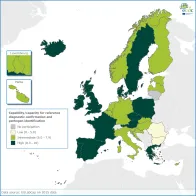
Availability of national reference laboratory testing capability and capacity and a robust sample referral and reporting system to the national authorities is a prerequisite for effective surveillance and epidemic preparedness at national and EU levels in accordance with NMFP consensus.

Development and implementation of harmonised methodologies to integrate molecular typing data into surveillance for priority diseases form a prerequisite for informing public health action based on EU-wide risk assessment of disease transmission.
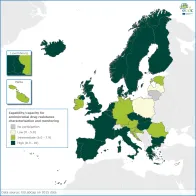
Accurate characterisation and monitoring of antimicrobial resistance determinants across human and animal populations for national/EU-wide surveillance informs public health action to contain cross-border and cross-species transmission of multidrug-resistant pathogens.

National surveillance networks connecting clinical/public health laboratories for reporting diagnostic information to surveillance databases and linking microbiological and epidemiological information are essential for efficient communicable disease and drug resistance surveillance and early infectious threat detection.
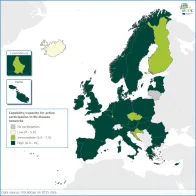
Active participation and collaboration between experts in EU disease networks promotes exchange of best practice and capacity building, which foster sufficient collective capacity in the EU for threat detection, investigation, disease surveillance and epidemic preparedness.

Preparation and involvement of the national reference laboratory capacities and staff in outbreak monitoring and response activities in collaboration with clinicians, epidemiologists, and microbiologists ensure the effective contribution of laboratory testing to support epidemic detection and control.
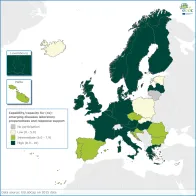
Up-to-date diagnostic capability for rare and (re)-emerging diseases and effective channels for collaboration are critical for laboratory preparedness and the deployment of timely and reliable emergency response to national and cross-border events.
Share this page
