ECDC publishes updated technical report on Options for the use of rapid antigen detection tests for COVID-19 in the EU/EEA
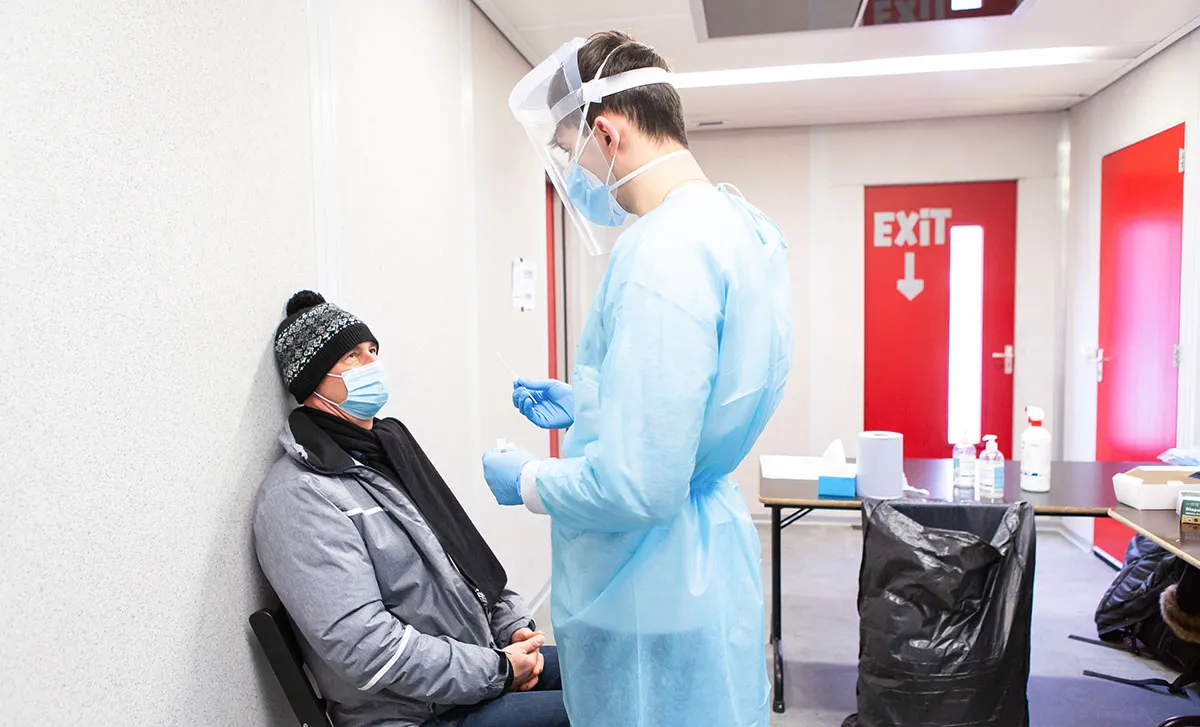
In the year since the original report was published, the overall epidemiological situation has continuously evolved, new variants have emerged, and vaccines have been deployed. This updated report is intended to facilitate further discussions between Member States with the aim of reaching agreement on the settings and purpose for which it is appropriate to use rapid antigen detection tests (RADTs) and summarises key considerations for their implementation.
Throughout the pandemic, testing for SARS-CoV-2 infection with nucleic acid amplification tests (NAAT) has remained the gold standard for detecting SARS-CoV-2. NAATs are characterised by both high sensitivity (the ability of a test to correctly identify those with the disease) and specificity (the ability of the test to correctly identify those without the disease) in detecting the presence of the SARS-CoV-2 virus. Many currently available RADTs show a lower sensitivity compared to the standard NAATs, while their specificity is generally reported to be high. It is important to note that RADTs are sensitive enough to detect cases with a high viral load. RADTs should be applied in a way that compensates for the lower sensitivity compared to NAAT, i.e. by including repeat testing for screening purposes and confirming test results by NAAT when appropriate.
RADTs can contribute to overall COVID-19 testing capacity and offer an advantage in terms of shorter turnaround time and reduced cost, especially in situations where NAAT testing capacity is reduced. The benefits of RADTs can help reduce transmission more efficiently through the timely identification of cases and faster contact tracing.
In February 2021 the Health Security Committee (HSC) established a list of mutually recognised RADTs for the purpose of issuing testing certificates together with the definition of clinical performance criteria. ECDC recommends Member States to use tests that are listed on the common list of COVID-19 RADTs as agreed by the HSC.
ECDC considers that appropriately validated RADTs, which are part of the HSC list, could be used to certify that a person has recovered from a past COVID-19 infection. However, self-test RADTs should not be used for issuing a formal certificate as proper sampling is crucial for diagnosis, and if testing is not performed correctly, a reliable test cannot be assured.
The emergence of virus variants triggered general concern about whether the analytical performance of RADTs could be affected by different virus variants. So far, the available data provide reassuring results for the use of RADTs to detect emerging virus variants.
Data describing the performance of RADTs in vaccinated individuals infected with SARS-CoV-2, irrespective of virus variants, have not yet been published.
Read the report

Share this page



