Technical guidelines on the prevention of communicable disease transmission through Substances of Human Origin
On 17 July 2024, the new Regulation (EU) 2024/1938 on standards of quality and safety for substances of human origin intended for human application was published. After entry into force, in August 2027 for most provisions, the regulation will repeal the Blood Directive (2002/98/EC) and the Tissues and Cells Directive (2004/23/EC). This regulation covers blood, tissues, cells, medically assisted reproduction, and other substances of human origin for human applications, with the exception of human organs for transplantation.
The SoHO regulation establishes ECDC as the expert body for developing and updating technical guidelines on the safety and quality of SoHO from a communicable disease threat perspective. The proposed Regulation stipulates that SoHO entities in the Member States shall take into account “the most recent technical guidelines, as indicated on the EU SoHO Platform […] published by the ECDC concerning the prevention of communicable disease transmission” (Article 56(4) and Article 59(4)).
In this context, ECDC is developing technical guidelines on the prevention of communicable disease transmission through SoHO in the European Union and European Economic Area (EU/EEA).
In the absence of technical guidelines published by ECDC, the safety requirements that apply are described on the page current safety requirements.
Published guidelines

Development of the technical guidelines
In June 2023, ECDC convened a first scientific expert panel to support ECDC in developing guidelines for the prevention of transmission of human immunodeficiency virus (HIV), hepatitis B virus (HBV), hepatitis C virus (HCV), T. pallidum, and the Human T-lymphotropic virus (HTLV). The work of this panel in providing scientific advice to ECDC is now concluded. The current timelines for the publication of these guidelines are described below.
Following the prioritisation exercise from the ECDC SoHO network, in January 2026, a new expert panel was convened to support ECDC in developing guidelines for relevant arboviruses, which include: West Nile virus (WNV), dengue virus (DENV), chikungunya virus (CHIKV), Zika virus (ZIKV), tick-borne encephalitis virus (TBEV), and Crimean-Congo hemorrhagic fever virus (CCHFV). This panel includes experts from the different SoHO areas (blood, tissues, cells, and medically assisted reproduction) and infectious disease specialists originating from 08 EU/EEA countries. The current timelines for the panel meeting and the development and publication of these guidelines are described below.
The European Directorate for the Quality of Medicines and Healthcare (EDQM) of the Council of Europe is represented by two observers in the scientific expert panel to avoid inconsistencies and gaps between the guidelines developed by ECDC and those developed by EDQM.
The European Medicines Agency (EMA) is also represented by an one observer in the scientific expert panel convened for arboviruses, in order to support a harmonised implementation of the technical guidelines that apply to plasma intended for industrial manufacturing.
ECDC provides the scientific evidence available in the form of evidence syntheses to support the expert panel discussions on relevant measures to prevent the transmission of pathogens through SoHO during the meetings of the panel. Outputs from the expert panel meetings will support ECDC in the development of technical guidelines on the prevention of communicable disease transmission through SoHO.
The technical guidelines developed by ECDC cover the testing and deferral strategies and laboratory testing methods for potential SoHO donors. According to the definition of SoHO in the regulation, these guidelines do not cover organ donors. In their first iteration, the guidelines will not cover breast milk or faecal microbiota.
Additionally, the first iteration of guidelines on HIV, HBV, HCV, T. pallidum, and HTLV does not cover plasma collected for industrial manufacturing. ECDC aims to adapt these guidelines to include this SoHO in the future and will provide relevant updates on this process on this webpage.
In the absence of technical guidelines published by ECDC for a given SoHO, the safety requirements that apply are described on the page current safety requirements.
Review and approval of the technical guidelines
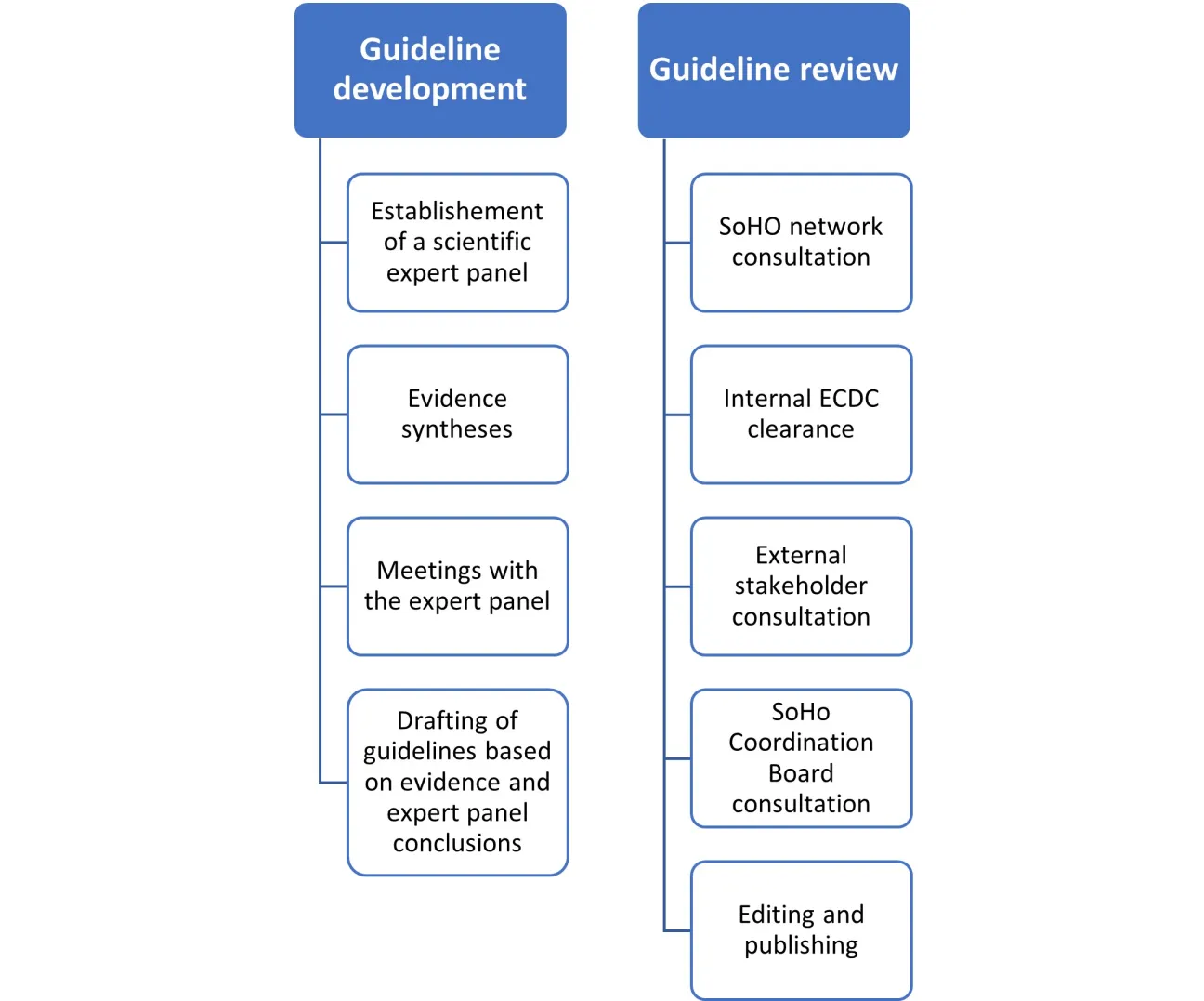
The ECDC SoHO network is consulted for the review of the draft technical guideline for a pathogen in the first step.
Following the ECDC SoHO network review and the internal ECDC scientific clearance, the technical guideline is sent for an external stakeholder consultation based on the list of stakeholder organisations established by the European Commission, as well as to EDQM, EMA, World Health Organization, and the SoHO Coordination Board (SCB). Organisations interested in reviewing the guidelines can register on the list of stakeholder organisations established by the European Commission.
The final guidelines will be published on the ECDC website and the EU SoHO platform described in the SoHO regulation.
Timelines for the development of guidelines
Timelines for the development of guidelines on HIV, HBV, HCV, T. pallidum, and HTLV – as of March 2026
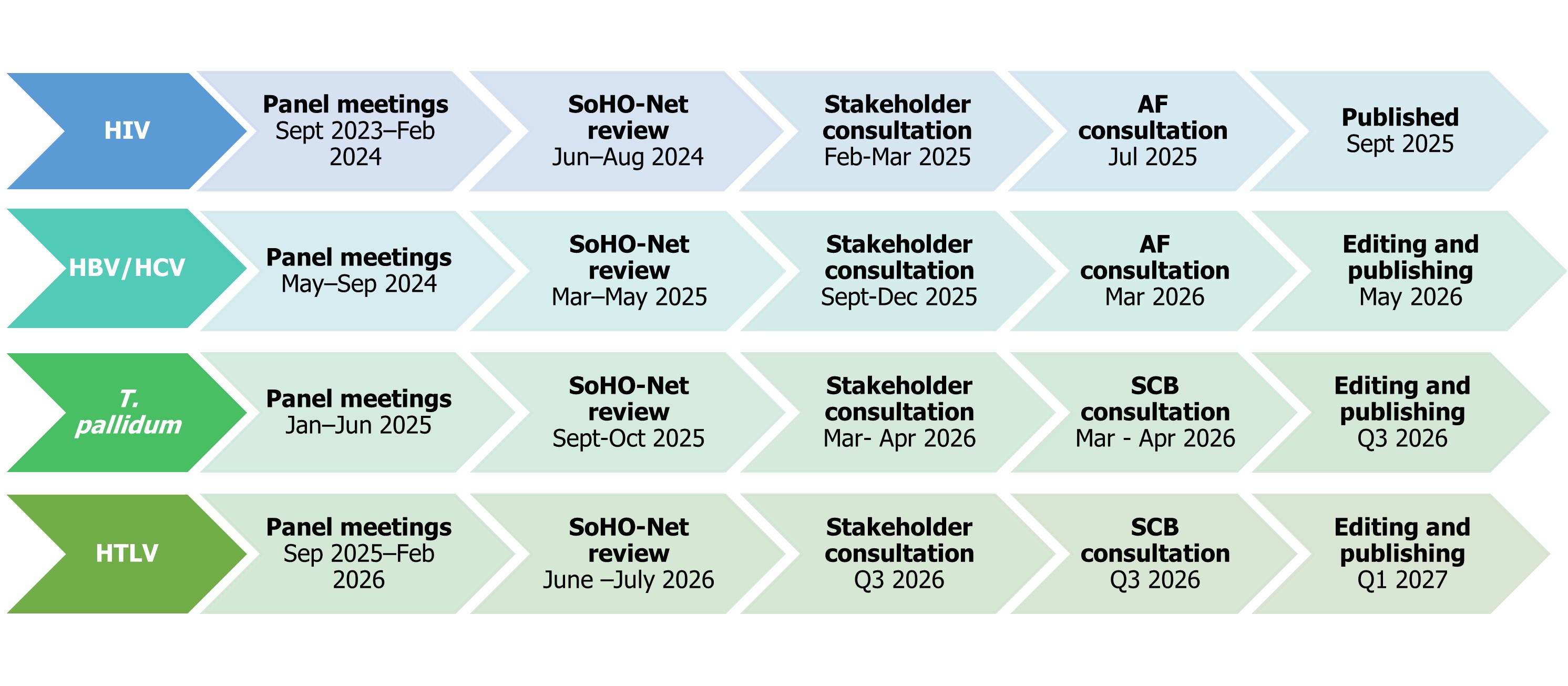
AF: ECDC Advisory forum; SCB: SoHO Coordination Board.
Note: the ECDC advisory forum consultation was replaced by consultation with the SCB for guidelines, with stakeholder consultation beginning at the start of 2026.
Timelines for the development of guidelines on DENV, CHIKV, ZIKV, WNV, TBEV, and CCHFV - as of March 2026
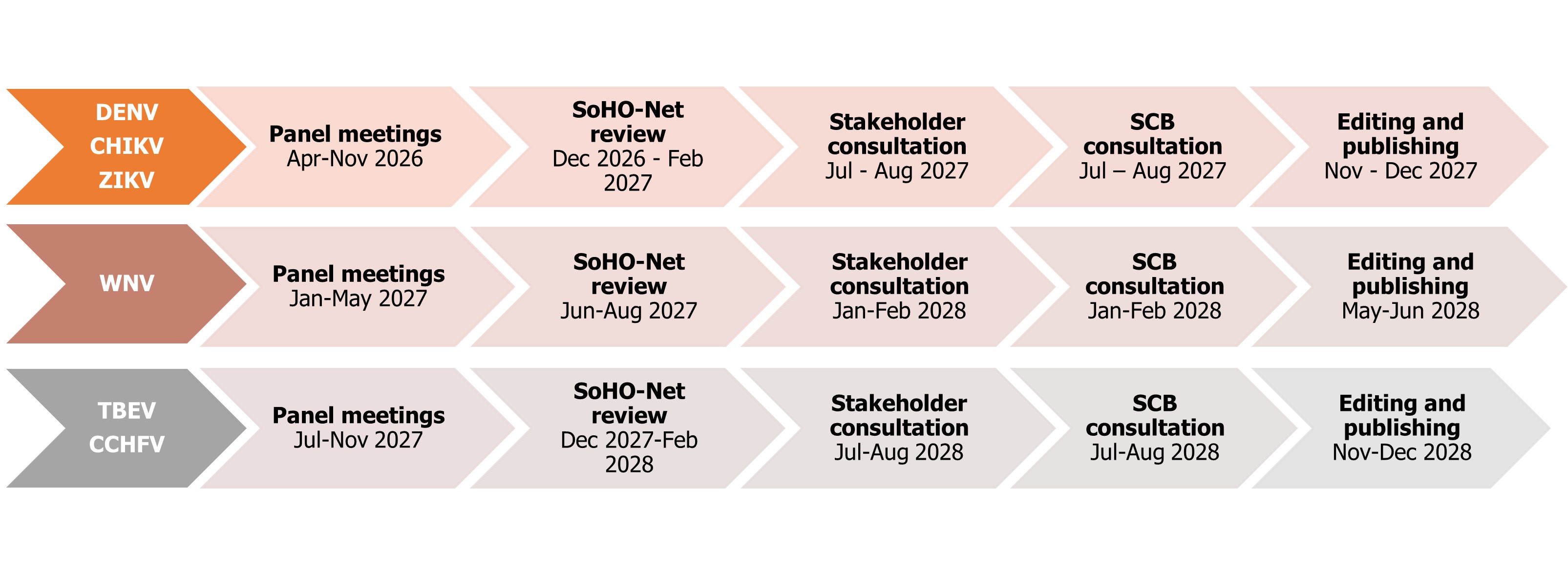
SCB: SoHO Coordination Board.
