Europe is steadily building more robust laboratory defences against epidemics and antimicrobial resistance threats
From the findings presented in the annual EULapCap report(link) published today it can be seen that Europe is steadily building more robust laboratory defences against epidemics and antimicrobial resistance threats. EU Member States showed strong capabilities with, as in previous years, use of harmonised methods for antimicrobial drug susceptibility testing. Indications, from the reductions of inequalities among EU Member States in laboratory capabilities, show a progress toward a stronger and more cohesive Europe for disease detection, surveillance and control.
Not all EU/EEA Member States, however, have reached sufficient levels of laboratory capability and capacity across all EULabCap targets, which are needed in order to deliver effective public health surveillance and threat response. With 19 of the 30 EU/EEA Member States in 2015–16 achieving ‘Sufficient microbiology capacity’ (defined as intermediate or high capacity for at least 10 of 12 EULabCap targets).
Areas that require further attention, to address insufficiencies, include:
- Development of wider clinical guidance for upgrading to genomic methods for the detection and characterisation of epidemic agents
- Guidance on the adequate utilisation of diagnostic tests
- Enhanced digital connections between laboratory information and public health monitoring and early warning systems at national and EU levels
However, the observable increases over the past four years suggests that, while there are current insufficiencies, public health microbiology shortcomings are being addressed. The narrowing variation in the EULabCap index between countries over the past several years indicates technical convergence and progress toward a more equitable balance of laboratory capacities among Member States.
These areas which were reviewed by ECDC, the competent bodies in the Member States, the European Commission, and several international partners in 2017 to inform the ECDC microbiology priorities and support activities for 2018–22.
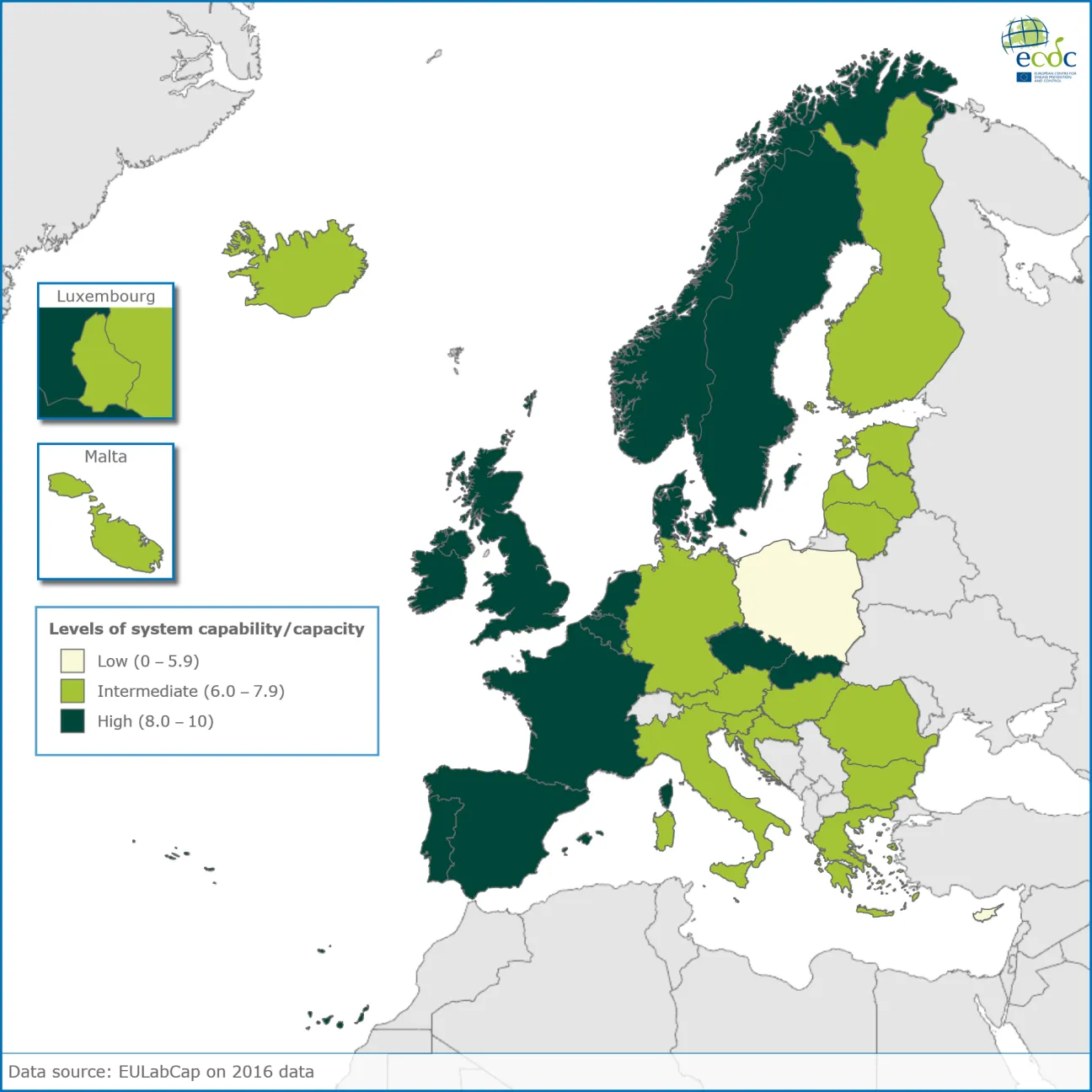
The EULabCap system
EULabCap (EU Laboratory Capability Monitoring System) is a tool for assessing and monitoring the laboratory capacities and capabilities in EU/EEA countries to underpin public health surveillance and assessment of risk posed by infectious disease. Furthermore, it gives an indication on the progression towards agreed upon practice standards and public health targets. This assessment aims at helping policy makers and practitioners identify possible areas for action and evaluate the impact of capacity strengthening activities and health system reforms. This report presents the indicator results of the 2016 data; country response rate to the 2016 survey was 100%. Data was provided for 97% of the applicable indicators (range per country, 90–100% complete data available). The average EULabCap 2016 index for all EU/EEA countries was 7.5 on a scale of 0–10, as compared to 7.5 in 2015, 7.3 in 2014, and 6.9 in 2013. In 2016, individual EULabCap indices per country ranged from 5.6 to 9.6 as compared to 4.7 to 9.2 in 2013, indicating that differences between national systems gradually decreased over the period 2013–2016. Ten countries improved their EULabCap index; five climbed from low to fair, and another five countries went from fair to high.
ECDC, in collaboration with the EU/EEA countries, the European Commission and other EU agencies and partners, will continue its monitoring of the European laboratory capacity as a basis for future country support and capacity building activities. The usefulness of the EULabCap monitoring system will be further evaluated by systematically collecting NMFP feedback on the use of reports for action at the national level. ECDC will also continue to appraise technological advances in microbiology, foster innovation, support the integration of harmonised genomic data into European surveillance systems, and share best practices across the European microbiology community.
Share this page

