Detailed data on COVID-19 vaccination progress in the EU/EEA available via Vaccine Tracker and in weekly reports
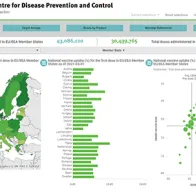
The new version of the Vaccine Tracker is now available as a separate dashboard. It provides information on the national uptake of the first and second doses of COVID-19 vaccines by age and other target groups, as well as by vaccine products, for all EU/EEA countries. Information is provided as maps, graphs and charts and can be exported as data.
Data provided in the Vaccine Tracker is the latest information reported to ECDC by the relevant authorities in EU/EEA countries through the EU surveillance system. Data are subject to retrospective corrections, and corrected datasets are released as soon as the processing of updated national data has been completed. ECDC works closely with all EU/EEA countries to continually improve data completeness and quality. For accurate interpretation of data, various factors affecting vaccine deployment in each country should also be considered. Monitoring will be further enhanced as more vaccines are rolled out and more information on vaccine coverage becomes available.
In addition to the latest data available in the Vaccine Tracker, the weekly COVID-19 vaccines roll-out overview report provides key figures and additional data as of the previous week. The latest weekly update indicates that, as of 14 February, nearly 22 million vaccine doses have been administered in the EU/EEA, and the estimated median uptake of the first vaccine dose among adults aged 18 years and above in the EU/EEA is 4.2% (among 29 countries reporting). Within the most vulnerable group – those aged 80 years and above – the estimated median uptake of the second vaccine dose (full vaccination) is 5.6% (among 20 countries reporting). These results should be interpreted with caution, as further work is ongoing to improve data quality and completeness.
Share this page