Factsheet on orthohantavirus infections
Case definition
Orthohantavirus infections can cause haemorrhagic fever with renal syndrome (HFRS), a clinical syndrome characterised by fever and acute kidney injury. HFRS is a notifiable disease in the EU under viral haemorrhagic fevers (VHF), and the EU case definition for surveillance of VHF is set out in Commission Implementing Decision (EU) 2018/945 of 22 June 2018 [1].
The pathogen
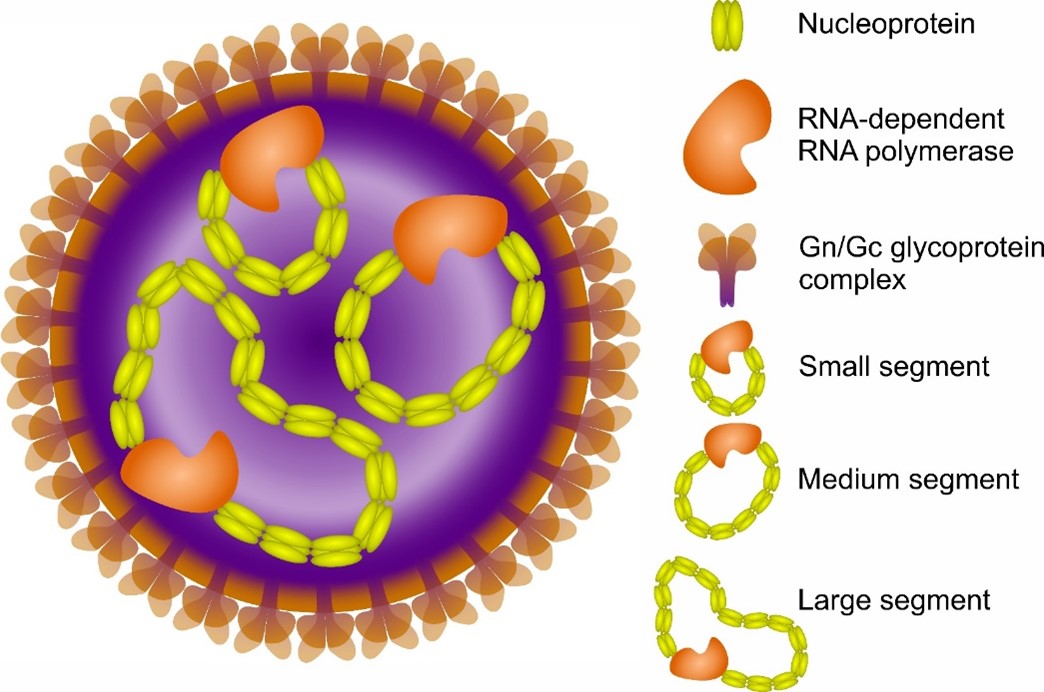
Orthohantaviruses (formerly known as Hantavirus genus) are enveloped, segmented, single-stranded, negative-sense RNA viruses belonging to the family Hantaviridae [1]. Members of Orthohantavirus genus are carried by rodents and include human pathogens [2]. The orthohantavirus genome encodes for nucleoprotein (NP), two glycoproteins (GP), and RNA-dependent RNA polymerase (RDRP) from its small (S), medium (M), and large (L) genome segments, respectively [1] (Figure 1).
Orthohantaviruses can cause severe disease in humans. In Eurasia, Dobrava virus (DOBV, Orthohantavirus dobravaense), Hantaan virus (HTNV, O. hantanense), Seoul virus (SEOV, O. seoulense) and other orthohantaviruses carried by mice and rats can cause HFRS. In Europe, Puumala virus (PUUV, O. puumalaense), carried by bank voles, typically causes a milder form of HFRS known as nephropathia epidemica (NE). Another vole-borne orthohantavirus distributed across Eurasia is Tula virus (TULV, O. tulaense), which can cause mild infections. In the Americas, Sin Nombre virus (SNV, O. sinnombreense), Andes virus (ANDV, O. andesense) and other orthohantaviruses carried by sigmodontine rodents can cause hantavirus cardiopulmonary syndrome (HPS) [4]. In Europe, human orthohantavirus infections are most frequently caused by PUUV, followed by low numbers of cases caused by DOBV [5]. Cases caused by SEOV and TULV have also occasionally been documented in Europe [6-10].
Clinical features and sequelae
The severity of HFRS varies and ranges from subclinical or mild to severe and fatal, depending on the causative orthohantavirus as well as the patient's immune response and genetic factors. The disease is primarily characterised by increased vascular permeability, hypotension, thrombocytopenia, and acute kidney injury (AKI) [3].
Traditionally, HFRS is divided into five phases: febrile, hypotensive, oliguric, polyuric, and convalescent. However, these phases can overlap, and it is often difficult to distinguish between them. The disease incubation period is around three weeks, and can range between 10 days and six weeks [4]. It typically begins with flu-like symptoms, such as high fever, headache, myalgia, nausea, and abdominal and back pain [3]. The febrile phase lasts around three to seven days and is often accompanied by sight/visual disorders. Visual/ocular symptoms are common during acute PUUV infection; in one prospective study, >70% of patients reported ocular symptoms, and myopic shift was frequently observed [5].
In the early acute phase, vascular leakage leads to the hypotensive phase, which can last from a few hours to two days. In severe cases, arterial hypotension and irreversible shock can occur. This phase is often accompanied by thrombocytopenia, which occurs in most HFRS cases, as well as coagulopathy and haemorrhagic manifestations, including petechiae on the skin and mucous membranes, haematemesis, epistaxis, melena, haematuria and, in severe cases, gastrointestinal bleeding and fatal intracerebral haemorrhage. Severe bleeding is rare in European patients with HFRS [3,4,6]. The oliguric phase usually lasts one to 16 days and is characterised by oliguria or anuria in around 30% of cases [4,7,8], with severe cases requiring dialysis treatment. AKI is common during this phase, with elevated serum creatinine and urea levels serving as important laboratory indicators.
In addition to renal damage, pulmonary, pancreatobiliary, central nervous system, endocrine and cardiovascular complications can also occur in some patients [8,9]. Electrocardiographic abnormalities, bradycardia, interstitial infiltrates in the lungs, pleural effusions and pulmonary oedema can also be observed in HFRS. The onset of the polyuric phase, which lasts from a few days to two weeks, is a positive prognostic sign indicating an improvement in renal function. The convalescence is usually prolonged and lasts up to six months. In Europe, between 30 and 50% of confirmed HFRS cases are hospitalised, with the most severe cases requiring intensive care [10]. In patients infected with DOBV, the disease is generally severe, with symptoms including ascites, pleural effusion, bleeding, oliguria/anuria and need of dialysis. The case fatality rate is 10–12% [11]. Infections with PUUV (leading to NE) are usually mild and have a case fatality rate of less than 1%.
HFRS can be lethal due to shock, complications of renal insufficiency and multiple organ failure. Most patients make a full recovery, and renal function is usually restored, although some studies suggest the development of hypertension in individual patients. Hormone deficiencies have been detected in 17% of patients (a median of five years) after PUUV infection [12]. Orthohantavirus infection can also increase the risk of stroke and heart attack [12-14].
Epidemiology
Members of the Hantaviridae family are known to be hosted by many different animals, such as rodents, shrews, bats, fish, and reptiles [1]. Of these hosts, only the viruses carried by rodents have been associated with human infections. Viruses in the genus Orthohantavirus are carried by rodents and can be genetically grouped to three main branches: i) viruses carried by Old World rats and mice (Muridae-borne); ii) viruses carried by voles and lemmings (Arvicolinae-borne); and iii) those carried by New World mice and rats (Sigmodontinae and Neotominae, also known as non-arvicolinae Cricetidae) [2]. Host rodents of murids (rodent species of Muridae family) are distributed across the globe, except for Antarctica. However, HFRS human cases have only been reported from Europe and Asia [15]. As with murid rodents, species of the Arvicolinae subfamily have spread widely across multiple continents, especially Europe, Asia, and North America. Orthohantaviruses carried by arvicoline rodents mostly cause NE [15], or they are non-pathogenic. NE cases are mostly reported from Europe [15].
Reported orthohantavirus human cases fluctuate throughout the years. These fluctuations can be driven by reservoir host abundance, climatic factors, or the frequency of human-host interactions [16-20]. In northernmost Europe, which has snowy winters, coniferous forests have multiannual predator-prey cycles with high bank vole abundancies in some autumns and early winters [18]. In Central Europe and the Balkans, climate-sensitive abundant seed crops of deciduous forests (masting) can result in high rodent densities over large areas [16,19]. These high rodent densities with increased abundances of orthohantavirus-infected rodents can lead to outbreaks and epidemic seasons of human orthohantavirus infections. PUUV prevalence in host rodents Clethrionomys glareolus (bank vole) can exceed 70% in outbreak settings [21]. The infection in the host rodent is persistent, so the host rodent has both the infectious virus and virus-specific antibodies while the host shows no signs of acute infection [22]. Once infected, the bank vole can shed the virus for the rest of its life [23,24].
In EU/EEA countries, reported human VHF cases caused by orthohantaviruses varies between about 1 500 and 5 000 cases annually [25]; Puumala virus (PUUV) is the most frequently identified cause of notified orthohantavirus infections. Due to typically mild symptoms, PUUV infection can go undiagnosed. As a result, PUUV infections can be more common than the notification rates suggest [26].
Transmission
The main route of transmission of orthohantavirus to humans is via inhalation of virus-contaminated aerosols from the urine, faeces or saliva of infected rodents. Although rare, rodent bites are also a possible route of infection [27]. As the transmission is thought to occur predominantly through the airways, smoking increases the risk, for example for PUUV infection about three-fold as compared to those who do not smoke [28]. Transmission risk is increased in association with outdoor activities (e.g. farming, forestry, gardening and recreational activities) or when cleaning rooms such as cellars and attics. Viremia can be short, but in severe cases can be prolonged [29].
Although PUUV RNA has been detected in the saliva of patients [30], human-to-human transmission has so far only been shown for ANDV, which is endemic in South America [31,32]. Vertical transmission during pregnancy has not been confirmed for DOBV or PUUV, although passive transmission of immunoglobulins has been observed [33]. However, transmission via breast milk has been reported for ANDV [34], which can also have the potential for sexual transmission [35].
Diagnostics
The clinical diagnosis of orthohantavirus infections can be done with collective observations made on symptoms and history of a possible contact with a rodent or travel history to areas where orthohantaviruses are known to occur in wildlife. Laboratory diagnosis of orthohantaviruses in patients is mostly carried out by serology (detection of acute-phase antibodies to orthohantaviruses), as patients are usually IgM-positive when they enter healthcare [36], but molecular methods (detection of viral RNA) are also useful in the first week of illness, especially in countries in which different orthohantaviruses are circulating [15].
Multiple assays are available both globally and in Europe where both acute (specific IgM antibodies) and past (with only IgG antibodies) infections can be detected [37] by using different serologic methodologies: enzyme immunoassay (EIA), immunofluorescence assay, immunoblot assay, and immunochromatography assay. Orthohantaviruses cross-react serologically if they belong to the same serogroup; these serogroups are similar to their genetic groupings explained above [15,37]. For diagnostic purposes, it is usually enough to diagnose an acute infection of a given serological group [37] (for endemic cases in Europe: either European PUUV-like or DOBV/SEOV-like; for imported cases: SNV/ANDV-type antisera cross-react with PUUV, and HTNV-type infections cross-react with DOBV/SEOV). Different tests can use different antigens or antigen pools in the assays. A specific distinction is possible by neutralisation tests but is rarely available or required for diagnosis. A study has suggested that serological assays based on antigen pools and lacking a specific identification of the causative orthohantavirus can provide false positives. In unclear cases, the use of a second confirmatory assay can enhance the accuracy of the serologic diagnosis [37]. The same study also mentioned that accuracy of serologic diagnosis of acute orthohantavirus infection is around 95%.
Molecular detection of orthohantaviruses from patients can be challenging, as the viremia can be short but is more sensitive than serology in early samples and in severe cases [15]. Whereas genus-specific (i.e. pan-hantavirus assay) tools can be applicable in rodents, virus-specific (i.e. DOBV- or PUUV-specific) polymerase chain reaction assays are more sensitive and are preferred for molecular detection in patients [37].
Isolation of orthohantaviruses is rarely successful from patients, but it can be done for research purposes, particularly from the rodent hosts. In the case of an isolation attempt, Vero E6 cell lines and serial passages have been used. However, the handling and isolation of viruses requires biosafety level (BSL) 2 for PUUV and BSL 3 for HFRS and HPS causing orthohantaviruses due to their risk classification under legal act EC Directive 2000/54/EC.
Case management and treatment
With the notable exception of ANDV, orthohantaviruses are not transmitted from person to person, so isolation of patients is not required.
There is currently no approved antiviral drug for orthohantavirus infections, and treatment is mainly symptomatic. Supportive treatment usually includes stabilisation of blood pressure with electrolyte infusions and hydration, and dialysis treatment for uraemia. The antiviral drug ribavirin has been used intravenously to patients with severe disease to reduce mortality and to prevent progression of oliguria and AKI [15]. However, a randomised, open-label study revealed insufficient efficacy and safety (i.e. adverse side effects) of ribavirin treatment in mild cases of PUUV infections [38]. In severe cases, where acute respiratory distress syndrome and severe AKI occur, mechanical ventilation and continuous renal replacement therapy might be required.
Research and development are ongoing, with therapeutic strategies such as antiviral drugs, immunotherapies and vaccine candidates being investigated. Icatibant acetate, a bradykinin receptor antagonist, has been used as a potential therapeutic option [39].
Public health prevention and control measures (for the authorities)
Prevention of human orthohantavirus infections focuses primarily on rodent control, avoiding contact with rodent excreta (such as urine, saliva or faeces) and minimising exposure to virus-contaminated dust [40]. Rodent control should include the elimination of food sources in and around the home, measures to prevent rodent entry, the use of rodent traps, and the removal of potential nesting sites near the home [41]. Other recommendations include cleaning areas contaminated with rodent droppings with disinfectants, ventilating rooms before entering them, and wearing rubber gloves and face masks to reduce the risk of exposure.
At the population level, an important preventive measure is the dissemination of information on risks of contact with the host rodent, modes of transmission, and personal protection strategies to residents and visitors in endemic areas. Communication strategies aimed at the general public and health professionals are crucial for prevention during non-epidemic periods and for a rapid response in the event of an outbreak
Contact tracing and active case finding of persons exposed to orthohantavirus are not considered necessary.
The response to an orthohantavirus outbreak depends on early identification of the pathogen and implementation of infection control measures involving laboratory, clinical, and public health personnel.
Infection control, personal protection and prevention (for the health facilities)
No vaccine against orthohantavirus infection or the associated diseases is currently authorised in the EU/EEA. Inactivated whole virus vaccines against HTNV or SEOV are used in South Korea and in China, but studies have shown they have a low level of efficacy [42].
Personal protective measures for people in at-risk groups (e.g. in occupations related to forestry, agriculture, military and outdoor activities) include avoiding contact with contaminated rodent excreta, wearing protective masks and gloves and using disinfectants when handling host rodents or being exposed to their excreta. PUUV has been shown to stay infective in room temperature for about two weeks and this is prolonged up to six weeks in cooler temperatures [43]. Laboratory work with orthohantaviruses should follow national guidance and pathogen-specific risk assessments.
References
- Bradfute SB, Calisher CH, Klempa B, Klingstrom J, Kuhn JH, Laenen L, et al. ICTV Virus Taxonomy Profile: Hantaviridae 2024. J Gen Virol. 2024 Apr;105(4) Available at: https://www.ncbi.nlm.nih.gov/pubmed/38587456
- Mull N, Seifert SN, Forbes KM. A framework for understanding and predicting orthohantavirus functional traits. Trends Microbiol. 2023 Nov;31(11):1102-10. Available at: https://www.ncbi.nlm.nih.gov/pubmed/37277284
- Vial PA, Ferres M, Vial C, Klingstrom J, Ahlm C, Lopez R, et al. Hantavirus in humans: a review of clinical aspects and management. Lancet Infect Dis. 2023 Sep;23(9):e371-e82. Available at: https://www.ncbi.nlm.nih.gov/pubmed/37105214
- Jonsson CB, Figueiredo LT, Vapalahti O. A global perspective on hantavirus ecology, epidemiology, and disease. Clin Microbiol Rev. 2010 Apr;23(2):412-41. Available at: https://www.ncbi.nlm.nih.gov/pubmed/20375360
- Hautala N, Kauma H, Vapalahti O, Mähönen SM, Vainio O, Vaheri A, et al. Prospective study on ocular findings in acute Puumala hantavirus infection in hospitalised patients. Br J Ophthalmol. 2011 Apr;95(4):559-62.
- Sundberg E, Hultdin J, Nilsson S, Ahlm C. Evidence of Disseminated Intravascular Coagulation in a Hemorrhagic Fever with Renal Syndrome—Scoring Models and Severe Illness. PloS one. 2011;6(6):e21134. Available at: https://doi.org/10.1371/journal.pone.0021134
- Turčinov D, Puljiz I, Markotić A, Kuzman I, Begovac J. Clinical and laboratory findings in patients with oliguric and non-oliguric hantavirus haemorrhagic fever with renal syndrome: an analysis of 128 patients. Clin Microbiol Infect. 2013 Jul;19(7):674-9.
- Pal E, Korva M, Resman Rus K, Kejzar N, Bogovic P, Kurent A, et al. Sequential assessment of clinical and laboratory parameters in patients with hemorrhagic fever with renal syndrome. PloS one. 2018;13(5):e0197661. Available at: http://www.ncbi.nlm.nih.gov/pubmed/29791494
- Vaheri A, Henttonen H, Mustonen J. Hantavirus Research in Finland: Highlights and Perspectives. Viruses. 2021 Jul 26;13(8) Available at: https://www.ncbi.nlm.nih.gov/pubmed/34452318
- Vaheri A, Henttonen H, Voutilainen L, Mustonen J, Sironen T, Vapalahti O. Hantavirus infections in Europe and their impact on public health. Rev Med Virol. 2013 Jan;23(1):35-49. Available at: https://www.ncbi.nlm.nih.gov/pubmed/22761056
- Papa A. Dobrava-Belgrade virus: phylogeny, epidemiology, disease. Antiviral Res. 2012 Aug;95(2):104-17.
- Mustonen J, Vaheri A, Porsti I, Makela S. Long-Term Consequences of Puumala Hantavirus Infection. Viruses. 2022 Mar 14;14(3) Available at: https://www.ncbi.nlm.nih.gov/pubmed/35337005
- Lupusoru G, Lupusoru M, Ailincai I, Bernea L, Berechet A, Spataru R, et al. Hanta hemorrhagic fever with renal syndrome: A pathology in whose diagnosis kidney biopsy plays a major role (Review). Exp Ther Med. 2021 Sep;22(3):984. Available at: https://www.ncbi.nlm.nih.gov/pubmed/34345266
- Connolly-Andersen AM, Hammargren E, Whitaker H, Eliasson M, Holmgren L, Klingstrom J, et al. Increased risk of acute myocardial infarction and stroke during hemorrhagic fever with renal syndrome: a self-controlled case series study. Circulation. 2014 Mar 25;129(12):1295-302. Available at: https://www.ncbi.nlm.nih.gov/pubmed/24398017
- Avsic-Zupanc T, Saksida A, Korva M. Hantavirus infections. Clin Microbiol Infect. 2019 Apr;21S:e6-e16. Available at: https://www.ncbi.nlm.nih.gov/pubmed/24750436
- Klempa B. Hantaviruses and climate change. Clin Microbiol Infect. 2009 Jun;15(6):518-23. Available at: https://www.ncbi.nlm.nih.gov/pubmed/19604276
- Reusken C, Heyman P. Factors driving hantavirus emergence in Europe. Curr Opin Virol. 2013 Feb;3(1):92-9. Available at: https://www.ncbi.nlm.nih.gov/pubmed/23384818
- Kallio ER, Begon M, Henttonen H, Koskela E, Mappes T, Vaheri A, et al. Cyclic hantavirus epidemics in humans--predicted by rodent host dynamics. Epidemics. 2009 Jun;1(2):101-7. Available at: https://www.ncbi.nlm.nih.gov/pubmed/21352757
- Tersago K, Verhagen R, Servais A, Heyman P, Ducoffre G, Leirs H. Hantavirus disease (nephropathia epidemica) in Belgium: effects of tree seed production and climate. Epidemiol Infect. 2009 Feb;137(2):250-6. Available at: https://www.ncbi.nlm.nih.gov/pubmed/18606026
- Buceta J, Escudero C, de la Rubia FJ, Lindenberg K. Outbreaks of Hantavirus induced by seasonality. Phys Rev E Stat Nonlin Soft Matter Phys. 2004 Feb;69(2 Pt 1):021906. Available at: https://www.ncbi.nlm.nih.gov/pubmed/14995490
- Tadin A, Bjedov L, Margaletic J, Zibrat B, Krajinovic LC, Svoboda P, et al. High infection rate of bank voles (Myodes glareolus) with Puumala virus is associated with a winter outbreak of haemorrhagic fever with renal syndrome in Croatia. Epidemiol Infect. 2014 Sep;142(9):1945-51.
- Vaheri A, Vapalahti O, Plyusnin A. How to diagnose hantavirus infections and detect them in rodents and insectivores. Rev Med Virol. 2008 Jul-Aug;18(4):277-88. Available at: https://www.ncbi.nlm.nih.gov/pubmed/18464294
- Voutilainen L, Kallio ER, Niemimaa J, Vapalahti O, Henttonen H. Temporal dynamics of Puumala hantavirus infection in cyclic populations of bank voles. Sci Rep. 2016 Feb 18;6:21323. Available at: https://www.ncbi.nlm.nih.gov/pubmed/26887639
- Voutilainen L, Sironen T, Tonteri E, Back AT, Razzauti M, Karlsson M, et al. Life-long shedding of Puumala hantavirus in wild bank voles (Myodes glareolus). J Gen Virol. 2015 Jun;96(Pt 6):1238-47. Available at: https://www.ncbi.nlm.nih.gov/pubmed/25701819
- Surveillance Atlas of Infectious Diseases. ECDC. 2026. Available at: https://atlas.ecdc.europa.eu/public/
- Latronico F, Maki S, Rissanen H, Ollgren J, Lyytikainen O, Vapalahti O, et al. Population-based seroprevalence of Puumala hantavirus in Finland: smoking as a risk factor. Epidemiol Infect. 2018 Feb;146(3):367-71. Available at: https://www.ncbi.nlm.nih.gov/pubmed/29310747
- Douron E, Moriniere B, Matheron S, Girard PM, Gonzalez JP, Hirsch F, et al. HFRS after a wild rodent bite in the Haute-Savoie--and risk of exposure to Hantaan-like virus in a Paris laboratory. Lancet. 1984 Mar 24;1(8378):676-7. Available at: https://www.ncbi.nlm.nih.gov/pubmed/6142362
- Vapalahti K, Virtala AM, Vaheri A, Vapalahti O. Case-control study on Puumala virus infection: smoking is a risk factor. Epidemiol Infect. 2010 Apr;138(4):576-84. Available at: https://www.ncbi.nlm.nih.gov/pubmed/19781114
- Korva M, Saksida A, Kejzar N, Schmaljohn C, Avsic-Zupanc T. Viral load and immune response dynamics in patients with haemorrhagic fever with renal syndrome. Clin Microbiol Infect. 2013 Aug;19(8):E358-66. Available at: http://www.ncbi.nlm.nih.gov/pubmed/23573903
- Pettersson L, Klingstrom J, Hardestam J, Lundkvist A, Ahlm C, Evander M. Hantavirus RNA in saliva from patients with hemorrhagic fever with renal syndrome. Emerg Infect Dis. 2008 Mar;14(3):406-11. Available at: https://www.ncbi.nlm.nih.gov/pubmed/18325254
- Martinez VP, Di Paola N, Alonso DO, Perez-Sautu U, Bellomo CM, Iglesias AA, et al. "Super-Spreaders" and Person-to-Person Transmission of Andes Virus in Argentina. N Engl J Med. 2020 Dec 3;383(23):2230-41. Available at: https://www.ncbi.nlm.nih.gov/pubmed/33264545
- Martinez VP, Bellomo C, San Juan J, Pinna D, Forlenza R, Elder M, et al. Person-to-person transmission of Andes virus. Emerg Infect Dis. 2005 Dec;11(12):1848-53. Available at: https://www.ncbi.nlm.nih.gov/pubmed/16485469
- Hofmann J, Fuhrer A, Bolz M, Waldschlager-Terpe J, Meier M, Ludders D, et al. Hantavirus infections by Puumala or Dobrava-Belgrade virus in pregnant women. J Clin Virol. 2012 Nov;55(3):266-9. Available at: https://www.ncbi.nlm.nih.gov/pubmed/22877560
- Ferres M, Martinez-Valdebenito C, Angulo J, Henriquez C, Vera-Otarola J, Vergara MJ, et al. Mother-to-Child Transmission of Andes Virus through Breast Milk, Chile(1). Emerg Infect Dis. 2020 Aug;26(8):1885-8. Available at: https://www.ncbi.nlm.nih.gov/pubmed/32687024
- Zust R, Ackermann-Gaumann R, Liechti N, Siegrist D, Ryter S, Portmann J, et al. Presence and Persistence of Andes Virus RNA in Human Semen. Viruses. 2023 Nov 17;15(11) Available at: https://www.ncbi.nlm.nih.gov/pubmed/38005942
- Kallio-Kokko H, Vapalahti O, Lundkvist A, Vaheri A. Evaluation of Puumala virus IgG and IgM enzyme immunoassays based on recombinant baculovirus-expressed nucleocapsid protein for early nephropathia epidemica diagnosis. Clin Diagn Virol. 1998 May 1;10(1):83-90. Available at: https://www.ncbi.nlm.nih.gov/pubmed/9646005
- Erdin M, Stanoeva KR, Mogling R, Korva M, Knap N, Resman Rus K, et al. External quality assessment of orthohantavirus and lymphocytic choriomeningitis virus molecular detection and serology in Europe, 2021. Euro Surveill. 2023 Oct;28(40) Available at: https://www.ncbi.nlm.nih.gov/pubmed/37796441
- Malinin OV, Platonov AE. Insufficient efficacy and safety of intravenous ribavirin in treatment of haemorrhagic fever with renal syndrome caused by Puumala virus. Infect Dis (Lond). 2017 Jul;49(7):514-20. Available at: https://www.ncbi.nlm.nih.gov/pubmed/28276794
- Vaheri A, Strandin T, Jaaskelainen AJ, Vapalahti O, Jarva H, Lokki ML, et al. Pathophysiology of a severe case of Puumala hantavirus infection successfully treated with bradykinin receptor antagonist icatibant. Antiviral Res. 2014 Nov;111:23-5. Available at: https://www.ncbi.nlm.nih.gov/pubmed/25194993
- European Centre for Disease Prevention and Control. Prevention measures and communication strategies for hantavirus infection in Europe. Stockholm: ECDC; 2014.
- Koivula TT, Tuulasvaara A, Hetemaki I, Makela SM, Mustonen J, Sironen T, et al. Regulatory T cell response correlates with the severity of human hantavirus infection. J Infect. 2014 Apr;68(4):387-94. Available at: https://www.ncbi.nlm.nih.gov/pubmed/24269676
- Liu R, Ma H, Shu J, Zhang Q, Han M, Liu Z, et al. Vaccines and Therapeutics Against Hantaviruses. Front Microbiol. 2019;10:2989. Available at: https://www.ncbi.nlm.nih.gov/pubmed/32082263
- Kallio ER, Klingstrom J, Gustafsson E, Manni T, Vaheri A, Henttonen H, et al. Prolonged survival of Puumala hantavirus outside the host: evidence for indirect transmission via the environment. J Gen Virol. 2006 Aug;87(Pt 8):2127-34. Available at: https://www.ncbi.nlm.nih.gov/pubmed/16847107
