EU Laboratory Capability Monitoring System (EULabCap): Report on 2014 survey of EU/EEA country capabilities and capacities
This report for 2014, which presents the indicator results for 30 EU and EEA (European Economic Area) countries, aims to help policymakers identify possible areas for action and to evaluate the impact of capacity strengthening activities and health system reforms.
Executive Summary
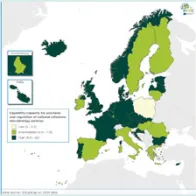
The results of the EU Laboratory Capability Monitoring System (EULabCap) survey on 2014 data confirms that the EU/EEA as a whole can rely on a strong public health microbiology system and substantial capacity to fulfil surveillance and response requirements. This second EULabCap report presents the results of the 2014 data and a comparison with the baseline of the first survey on 2013 data for 30 EU/EEA countries.
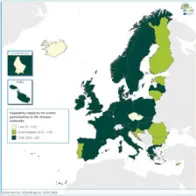
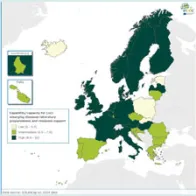
There are still substantial differences in EULabCap indices between the EU/EEA countries but many countries have showed early indications of an improved system capability and capacity in 2014 compared to the previous year. In particular, there is a clear progress in the monitoring of antimicrobial resistance and support of national reference laboratories in outbreak response. Another notable development is that eight EU/EEA countries have started in 2014 to use whole genome sequencing (WGS)-based typing for the routine surveillance of at least one disease. This technology shift is in line with the ECDC strategy on use of WGS for public health applications. ECDC is supporting the Member States in the transition to WGS from earlier typing technologies to enhance national and EU-level surveillance.
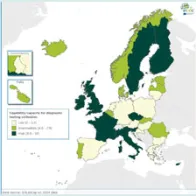
In the EULabCap 2013 survey, there were limited requirements for accreditation and licencing of clinical microbiology laboratories, which are processes to ensure the reliability of results in diagnosis and treatment of patients. Progress was noted in the 2014 survey with more countries requiring a licencing authorisation/registration of their laboratories. However, gaps remained in biosafety regulation and oversight which reinforce laboratory containment of infectious microorganisms and hazardous biological materials.
laboratory-capability-monitoring-2014-eu-labcap.pdf
English (13.05 MB - PDF)



Share this page